Associations between obstructive sleep apnoea and the development and severity of retinal vein occlusion
In this study comparing a large cohort of patients with and without a recorded ICD code for OSA among various racial/ethnic and sex subgroups, OSA was found to be associated with an increased risk of having a new ICD code of RVO in both males and females, and NHW, Black, and H/L population. These findings are consistent with those of existing studies [3,4,5, 11] that have investigated the prevalence of RVO among patients with OSA in the general population. Moreover, even among those with preexisting ICD codes for RVO, those with OSA had an increased risk of being diagnosed with vision-threatening complications and of undergoing invasive treatment procedures.
Although the current literature lacks specific data on how sex influences the relationship between OSA and RVO, epidemiological studies consistently show that males are at a significantly higher risk of developing OSA than females [1, 10]. While one might expect that OSA’s association with RVO risk would also differ by sex, our stratified analysis revealed that OSA is associated with an increased risk of being diagnosed with RVO equally in both males and females. This suggests that sex does not significantly influence the association, indicating that other factors are likely driving the positive association between OSA and the risk of RVO. One such factor to consider is the presence of metabolic comorbidities, including hypertension, hypercoagulability, and atherosclerosis, which are well-established risk factors for RVO [12, 13]. While all cohorts in this study were matched for the presence of such baseline comorbidities via propensity score matching, the severity of such risk factors could not be fully controlled. Therefore, possible explanations for the observed findings are that patients in the OSA cohort had more severe metabolic conditions that naturally increased their risk of being diagnosed with RVO, or that OSA served as a synergistic risk factor that aggravated the effect of these comorbidities on the development of RVO. Replication of the findings of this study after a more granular assessment of the severity of these comorbidities, as well as additional exploration of other potential mediating factors, is necessary to confirm these hypotheses and further elucidate the mechanisms underlying the relationship between OSA and RVO. In addition, as CPAP therapy has been shown to significantly reduce the severity of metabolic conditions in patients with OSA [14,15,16] and prevent maculopathy among patients with diabetes [17, 18], whether early initiation of CPAP therapy can effectively mitigate the risk of RVO in patients with OSA would be a valuable area for further research.
Our race/ethnicity-stratified analysis showed that, among NHW, H/L, and Black populations, OSA is associated with a higher risk of having a new diagnosis of BRVO, but not CRVO. While both types of RVO share several risk factors [19], some studies have also highlighted nuanced differences in the degree to which each risk factors affect BRVO and CRVO. For instance, hypertension, vascular disease, and thrombotic conditions—all of which are predominant complications of OSA—have shown to have stronger association with BRVO than CRVO [20, 21]. Conversely, some studies have demonstrated that the prevalence of OSA is higher in patients with CRVO than those without [22, 23]. As the differences in the pathogenesis of BRVO and CRVO are still incompletely understood, further research is warranted.
When comparing various racial/ethnic groups, we found that having an ICD code for OSA was associated with the highest risk of receiving a new ICD code for RVO in the H/L population. One previous study comparing the overall prevalence of RVO among White, Black, Hispanic, and Chinese Americans found that race or ethnicity was not a significant risk factor for RVO development [24]. This study, however, excluded patients with cardiovascular diseases, which is one of the most significant risk factors of RVO20, and a pooled analysis of multiracial studies by Rogers et al. later showed that the age- and sex-standardized prevalence of RVO was indeed highest in H/L patients [25]. Currently, there is insufficient evidence to determine whether H/L ethnicity itself is an independent risk factor for RVO development or if it predisposes individuals to a greater detrimental effect of OSA on RVO risk, particularly as the higher rates of socioeconomic health hazards faced by H/L populations compared to other groups [26]. Regardless, the results of our study highlight the importance of focusing on H/L patients in future investigations of the relationship between OSA and RVO, especially given the significantly lower representation of H/L populations in existing clinical trials on RVO [27].
Conversely, there was no association between OSA and the incidence of RVO among Asian patients. This finding is notable as Asians are at higher risk for severe OSA and its complications—even at lower BMI compared to other racial groups—due to anatomical differences in craniofacial and upper airway structures [28,29,30]. Furthermore, while no published studies have compared Asians with other racial groups specifically on the impact of OSA and RVO, a nationwide study in Taiwan showed that the incidence of RVO was higher among OSA patients compared to controls [3, 4, 31]. The contrasting finding of this study may be explained by the heterogeneous makeup of the Asian American population, which includes not only East Asians (e.g. China, Taiwan, Hong Kong, Korea, Japan, Mongolia) but also South Asians, Southeast Asians, and others, who vary widely in their craniofacial anatomy, obesity rates, diets, and healthcare access [32,33,34,35]. A relative underdiagnosis of OSA among Asian Americans may have influenced the power of this study to detect the association between OSA and RVO, as supported by both the literature and the relatively low sample size of the Asian subgroup compared to other racial and ethnic subgroups in this study [36].
Finally, our analysis of patients with preexisting RVO suggested a positive association between OSA and the risks of having more severe RVO or complications, including ME, VH, NV, and the need for invasive treatment via PRP and intravitreal injections. Similarly, Wan et al [4]. reported that RVO patients with OSA have a higher risk of developing MO and have limited improvement in post-treatment visual acuity. Although the risk of NV was not significantly associated in their study (possibly due to limited follow-up duration) [4], the association between NV and OSA seen in our study is consistent with the well-established understanding that recurrent episodes of nocturnal hypoxia and reperfusion in OSA lead to pathological production of VEGF and oxidative stress that leads to endothelial dysfunction [4, 37, 38]. Besides, OSA has been implicated in inducing additional changes in the retinal veins, including increased tortuosity [39], decreased vessel density [40], and narrowing [41], all of which can render patients more vulnerable to clinical progression. This suggests that regular monitoring of patients with RVO with concurrent OSA and prompt treatment of OSA may play a vital role in preventing vision-threatening complications of RVO and reducing treatment burdens.
Limitations
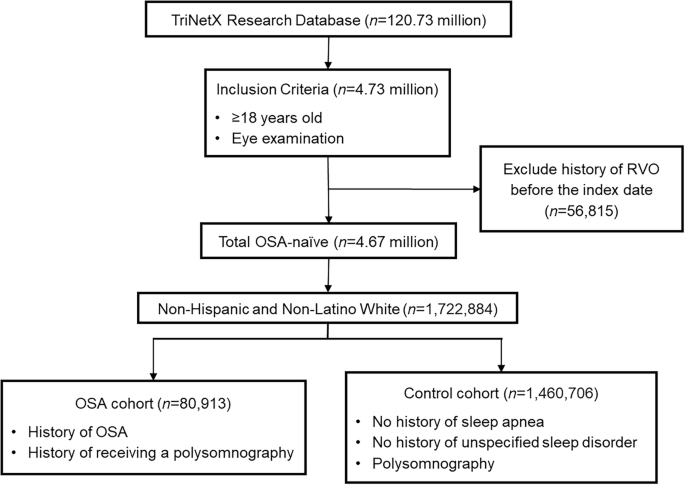
As a retrospective study that relied on EHR data, this study has limitations that must be acknowledged. While we strived to increase the accuracy of the OSA diagnosis by adding polysomnography testing into our inclusion criteria, detailed results of polysomnography, such as apnoea-hypopnea index (AHI) and desaturation levels, were not available. Therefore, we cannot definitively conclude that the patients in the OSA cohort were diagnosed based on objective polysomnography criteria. Similarly, the variability in OSA severity within the OSA cohort may have confounded our results. Additionally, as OSA can be significantly underdiagnosed, misclassification bias could have skewed our results to significantly underrepresent the true association between OSA and RVO.
In addition, although we included CPAP therapy as one of the covariates, we could not standardize patients’ level of adherence, duration, and responses to CPAP therapy. Consequently, variability in such factors could have additionally confounded our results. Given the relative deficiency of data on the impact of CPAP therapy on RVO, controlled trials in this area would help verify the positive associations between OSA and RVO observed in this study and previous studies and potentially offer an avenue to prevent RVO development and exacerbation.
While our large sample included patients from various racial and ethnic backgrounds, Asian patients were underrepresented. As such, it is possible that the absence of an association between OSA and RVO among Asian patients in this study could be due to underpowered analysis. Furthermore, we could not separately analyse American Indian, Alaska Native, Native Hawaiian, or other Pacific Islander populations due to sample size limitations.
Because the risk of RVO is multifactorial, unmeasured differences between the cohorts—such as in socioeconomic status, lifestyle habits, and other cardiovascular risk factors not included in the PSM—may have affected the results of our analysis. Although this study employed PSM to minimize such impact, residual effects were likely because medical record data on lifestyle habits, social determinants of health, and health-seeking behaviours are often incomplete and underreported [42, 43].
Finally, this study performed a sensitivity analysis to strengthen the validity of our analysis of patients with preexisting RVO by excluding patients with potential confounding ocular conditions. While the significantly higher proportion of events (complications or invasive treatment for RVO) in the OSA cohort supports the results of the primary analysis, the reported RRs in this sensitivity analysis may not be exact, as fewer than 10 controls had events (Table 2A) and TriNetX reports any counts of 1 to 10 as 10 to protect patient privacy.
link